Empower
How We Help
According to Tufts CSDD, decentralized and hybrid trials finish 2-3 months faster with a net benefit up to 14x greater than the up-front technology investment.
- Make it easy for patients to participate
- Improve medication compliance
- Increase patient retention, convenience, and safety
Patient Ride
Up to 50% of patients cite transportation challenges as a reason not to enroll in a clinical trial. That figure only increases for:
Therapeutic areas such as Oncology where patients are physically burdened or exhausted
Patients who have financial hardship or rely on caregivers for transportation
Lengthy studies or those with many site visits
Trials with procedures that limit or restrict patients from driving
Thankfully, ClinOne has partnered with Uber Health to integrate reliable transportation options directly into our single platform.
With ClinOne’s Patient Ride solution, patients and site staff can hail or schedule Uber Health rides to/from site visits or other approved destinations – with all billing centralized and managed by ClinOne within your study budget. This relieves the burden on patients, increases enrollment and retention, and reduces the burden on sites.
Read this case study to see how one sponsor provided more than 1,250 patient rides in a single clinical trial.

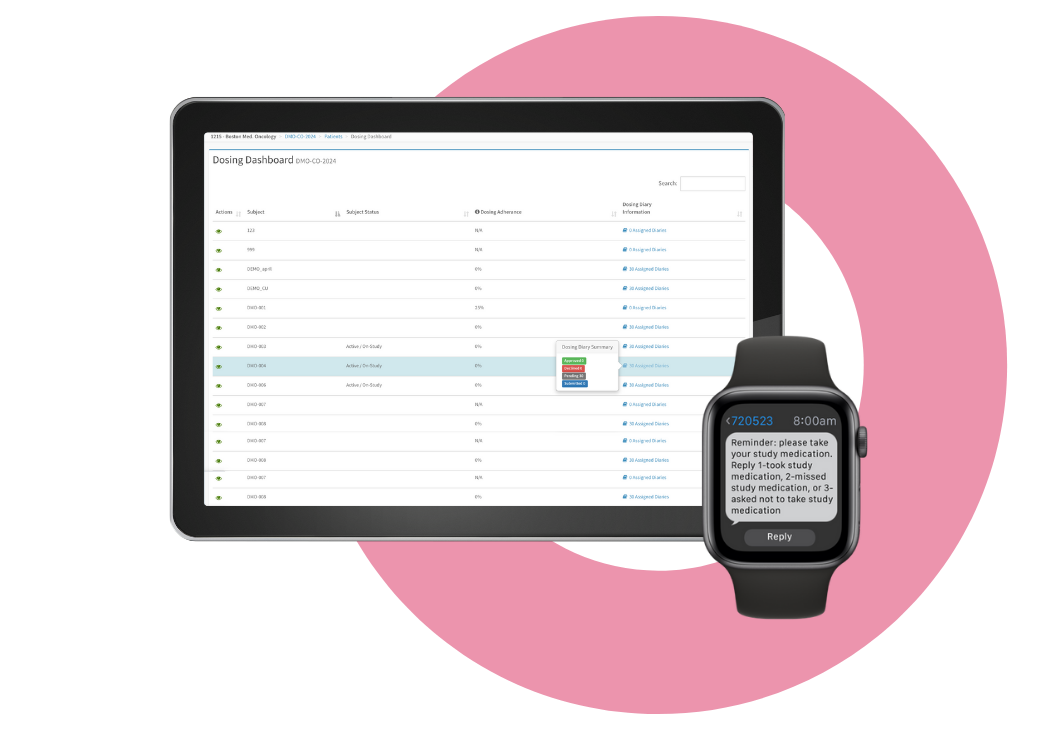
Medication Adherence
On average, 40% of clinical trial patients become non-compliant with self-medication protocols at 5 months of study, and it only gets worse from there. That’s why our single platform includes a Medication Adherence solution that uses interactive SMS to send medication reminders and receive confirmation from patients’ mobile phones.
As a result, one ClinOne customer achieved compliance as high as 98% compliance at 18 months of study – with an elderly population and a twice-daily dosing requirement.
Whether it is for your study drug or background medications, ClinOne can help make sure your patients are compliant, so you can get a true assessment of how your treatment is working.
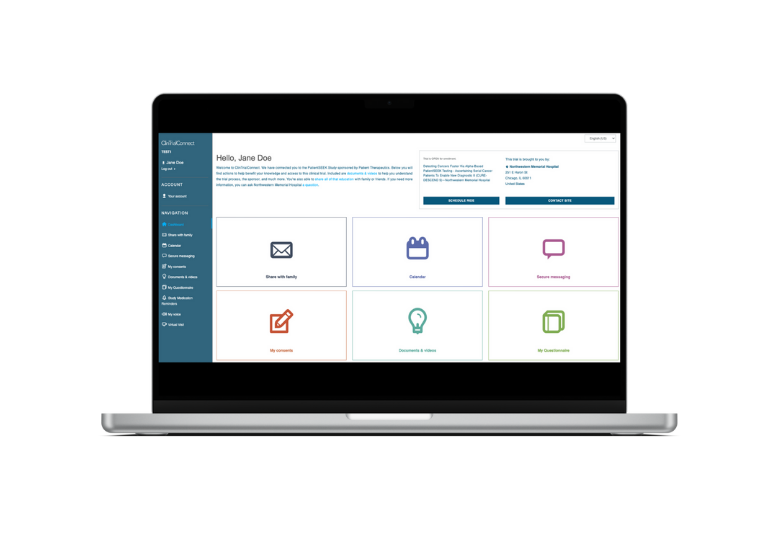
Patient Engagement
One positive to come out of the global pandemic was the acceleration of the adoption of clinical trial technology solutions aimed at engaging patients where they are, which has helped expand access to patients in more remote areas and support diversity in clinical research. We explore this topic in detail in our white paper “The Evolution of Patient Technology in Cancer Clinical Research”.
In addition to Patient Ride and Medication Adherence, our Patient Engagement Portal includes:
Video visits for up to 10 participants
Enterprise Consent Management
Quality of Life eDiary
Secure site and patient messaging
Visit scheduling using patients’ calendar of choice
Caregiver support and sharing with family
Digital concierge and trial resources
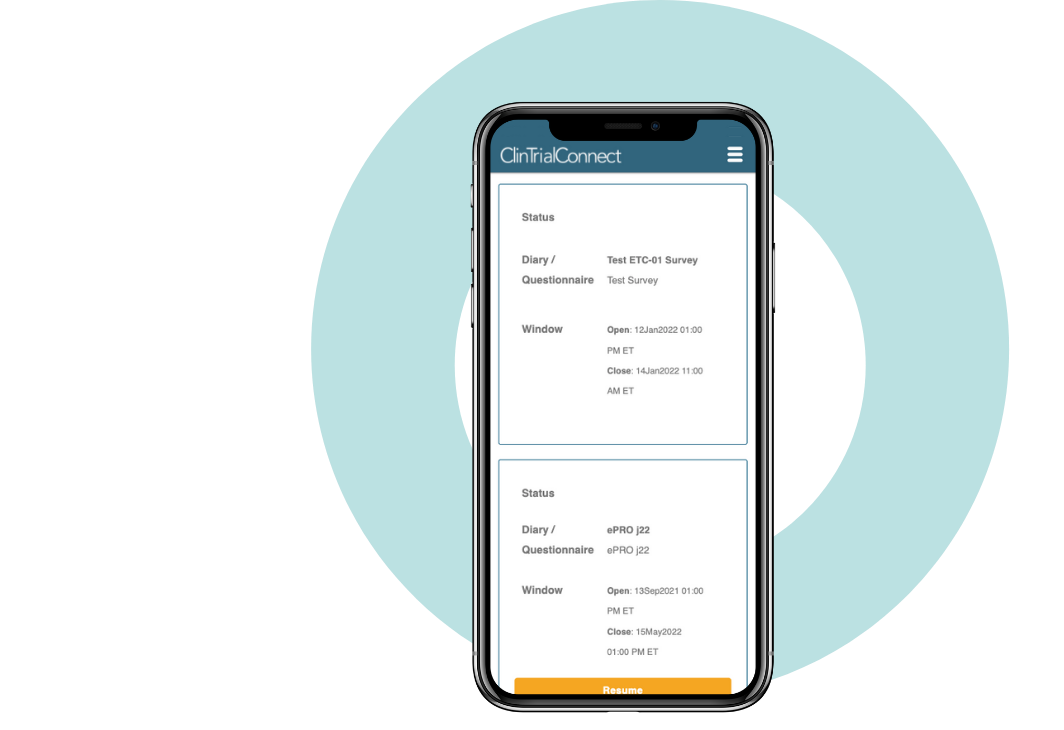
eDiary
If you want to know how patients are feeling during your clinical trial – why not ask them?
Unfortunately, paper diaries have been proven time and again to be unreliable with failures including:
Access to data is delayed by weeks, preventing proactive site follow-up, adaptive trial designs, or interim analyses
Incomplete, illegible, and illogical responses with no ability for data quality edit checks
Entries are not time-stamped, inviting recall bias and the ‘parking lot syndrome’
Makes it easy for you to capture trustworthy quality of life (QoL) data
This introduces tremendous risk and hidden costs such as poor data quality requiring over-enrollment to compensate for lower study power, extra site monitoring expenses with numerous queries to resolve, and significant delays. Thankfully, for these reasons and more, the industry has largely moved on to electronic patient diaries.
As a BYOD solution, ClinOne eDiary makes it easy for you to capture trustworthy quality of life (QoL) data and other self-reported information, which are increasingly being used for labeling decisions – especially in Oncology where payers want to see a QoL improvement for a drug that has similar therapeutic benefit to existing treatments.
Our eDiary solution is simple and familiar for patients using their own mobile devices or internet browsers of choice. It is an integral part of our true single platform which has been deployed for patients in 60+ native languages and 55+ countries around the world.
True Single Platform
ClinOne is a true clinical research platform aimed at offering an Adaptive Experience to all participants. As opposed to products cobbled together from disparate systems acquired by M&A, we put the features sites, patients, and caregivers need in one place at their fingertips with a single user experience – helping them actively participate in your remote, on-site, or hybrid trial.
